 In August 2013, our interdisciplinary research team was awarded a Major Instrumentation-Development grant from the National Science Foundation to develop an integrated instrument for small animal hyperthermia research under ultra-high field magnetic resonance imaging (MRI). The principal investigator of the project is Dr. Stefan Bossmann (Chemistry), with the instrument design, development, and integration aspects led by Dr. Punit Prakash (ECE). Other co-PIs on the project are Dr. Leila Maurmann (Chemistry), Dr. Sanjeev Narayanan (Diagnostic Medicine/Pathobiology), and Dr. Deryl Troyer (Anatomy and Physiology). Dr. Sergio Curto (ECE) is spearheading the implementation and evaluation of MRI-compatible hyperthermia applicators.
In August 2013, our interdisciplinary research team was awarded a Major Instrumentation-Development grant from the National Science Foundation to develop an integrated instrument for small animal hyperthermia research under ultra-high field magnetic resonance imaging (MRI). The principal investigator of the project is Dr. Stefan Bossmann (Chemistry), with the instrument design, development, and integration aspects led by Dr. Punit Prakash (ECE). Other co-PIs on the project are Dr. Leila Maurmann (Chemistry), Dr. Sanjeev Narayanan (Diagnostic Medicine/Pathobiology), and Dr. Deryl Troyer (Anatomy and Physiology). Dr. Sergio Curto (ECE) is spearheading the implementation and evaluation of MRI-compatible hyperthermia applicators.
Hyperthermia is a treatment where localized regions of the body are heated to temperatures in the range of 40-44 °C. Several clinical trials have shown the benefits of moderate heating within this temperature range to augment the efficacy of radiation therapy and chemotherapy of cancer. Hyperthermia also stimulates a mild anti-tumor immune response and facilitates highly localized heat-triggered release of therapeutic agents. MRI affords non-invasive, real-time monitoring of the spatial temperature profile induced in tissue during heating. As such, it provides a means for real-time monitoring of the progression of hyperthermia procedures, and may be integrated with feedback control techniques based on thermal, anatomic, and/or physiologic changes in tissue during treatments.
The goal of our project is to design an instrument that facilitates the design and execution of pre-clinical hyperthermia experiments in small animals under ultra-high field (14 T) MRI guidance. For comparison, most scanners in current clinical use operate at 1.5 T or 3 T. After considering small-animal MRI technologies available from several companies, our group decided to acquire a Bruker 14 T (600 MHz) Avance III scanner. This custom instrument was designed in Switzerland and was installed at KSU in Fall 2014. We anticipate that the entire instrument including an integrated image-based feedback control system will be ready for use by researchers in Spring 2016.
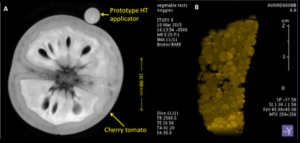
When complete, this instrument will facilitate real-time imaging of changes induced by heating at an unprecedented spatial scale (on the order of ~10 μm). Our approach includes a custom-designed microwave antenna, made of MRI compatible materials, that directs energy towards the targeted tissue within the experimental small animal. A prototype microwave hyperthermia applicator has been fabricated and experiments have demonstrated MRI compatibility with minimal imaging artifacts introduced by the applicator. Computational electromagnetics and bioheat transfer simulations have been employed to characterize the performance and of the applicator, and efforts are underway to integrate the applicator within the MRI environment. This project has provided an excellent opportunity for ECE research students to develop and apply approaches from electromagnetics and antenna design, computational modeling, image processing, and control algorithms to a healthcare problem.
We plan to use this system to investigate the use of hyperthermia and heat-triggered release of chemotherapeutic and immune-stimulatory agents for targeting pancreatic cancer and localized infections in small-animal models. We anticipate this resource to be of interest to other researchers in the region interested in investigating the physical, anatomic, and physiologic changes induced by interventional devices and treatments under ultra-high field MRI.