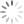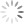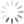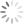(Megan Kennelly, KSU Plant Pathology)
Around this time every year we all look forward to early September. Often Labor Day weekend marks the end of “summer diseases” and the beginning of fall repair and renovation to our cool-season grasses.
Moisture and humidity continue to trigger turf diseases. Here are a few recent photos:
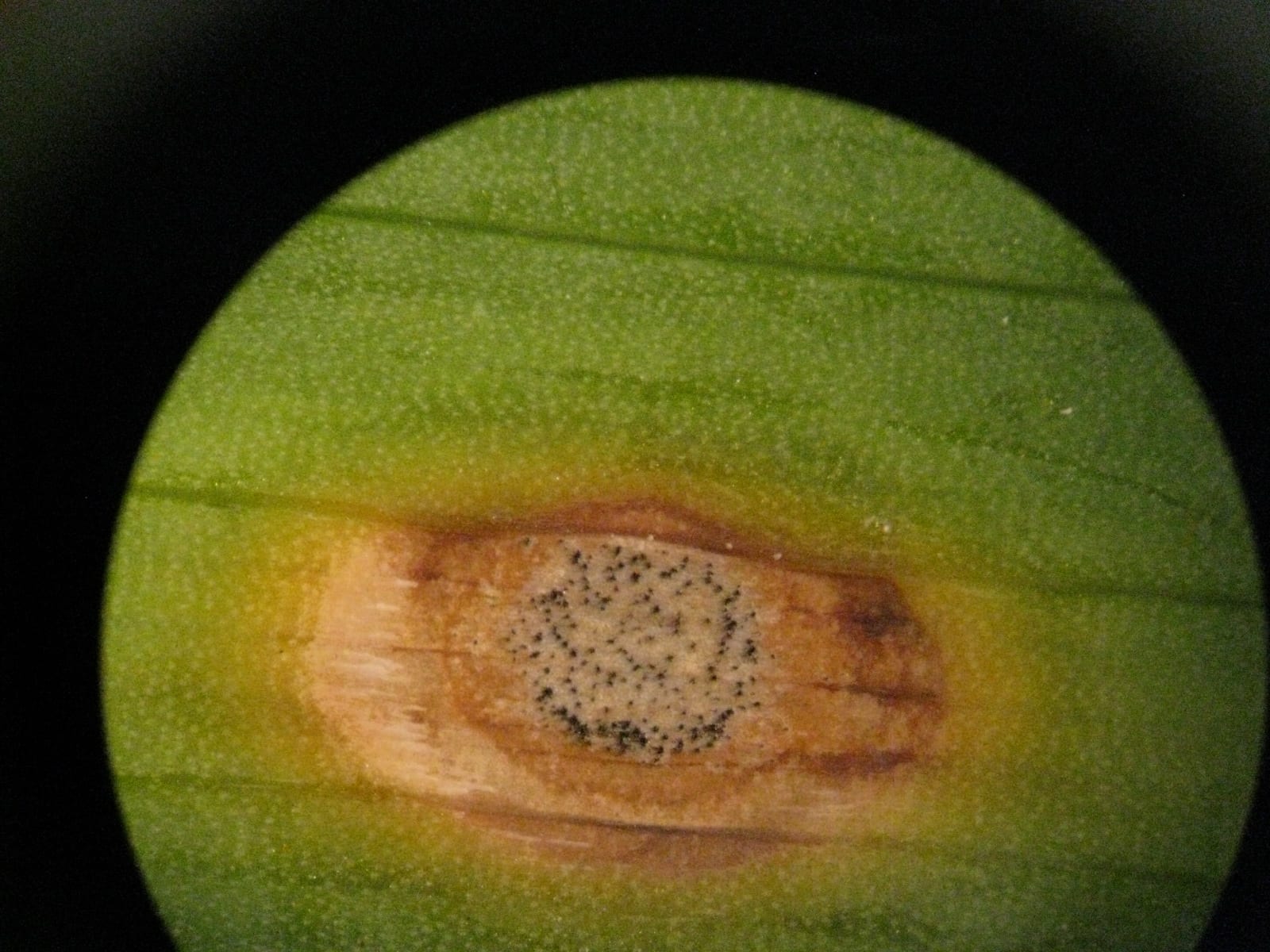
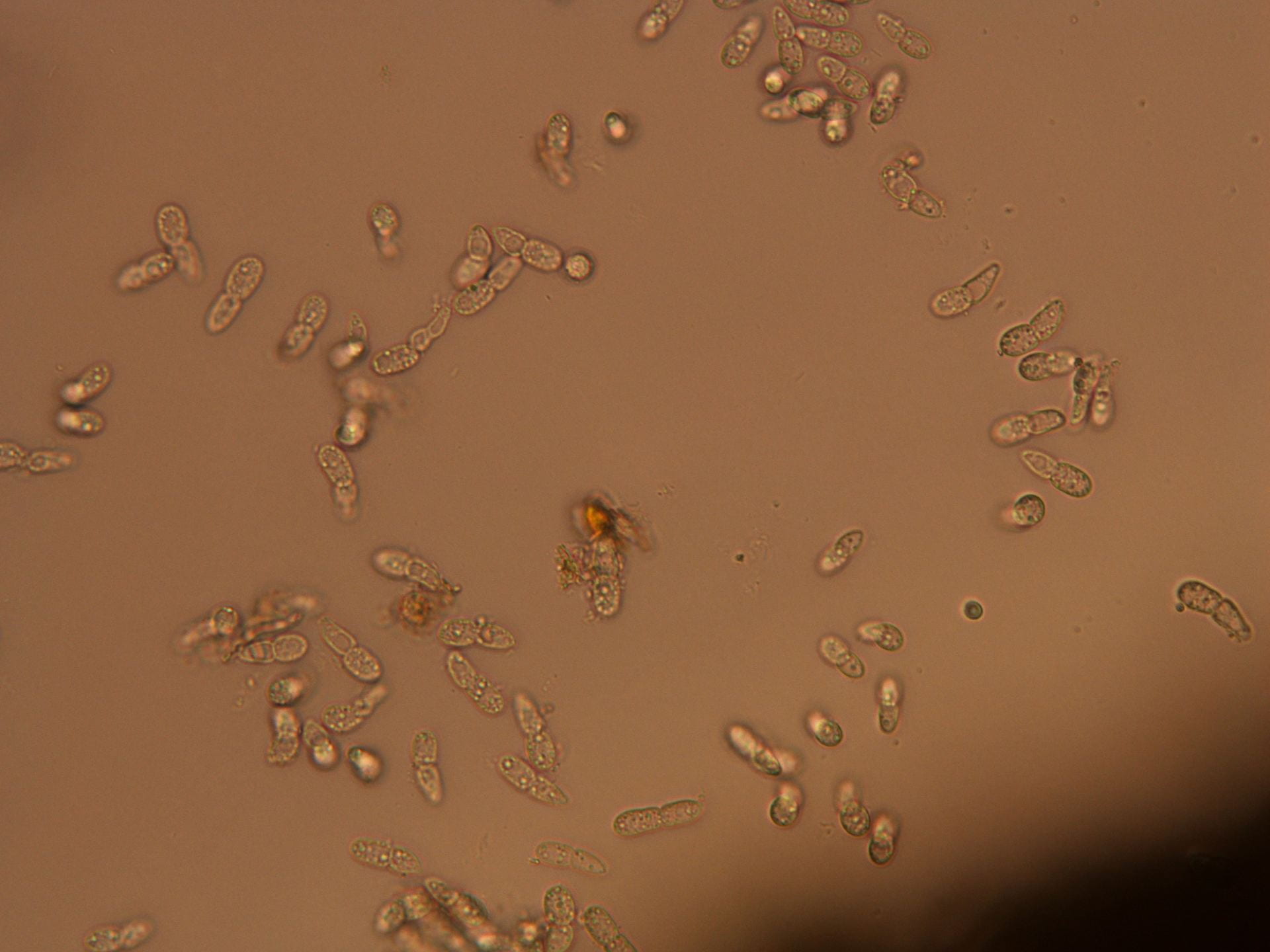
Brown patch
Brown patch in tall fescue, with mycelium present on a wet, humid morning. The first photo shows the characteristic lesions. As I write this on Friday Aug 16 there is a stretch of hot days and very warm nights ahead, so this disease may continue. But September is not far away…


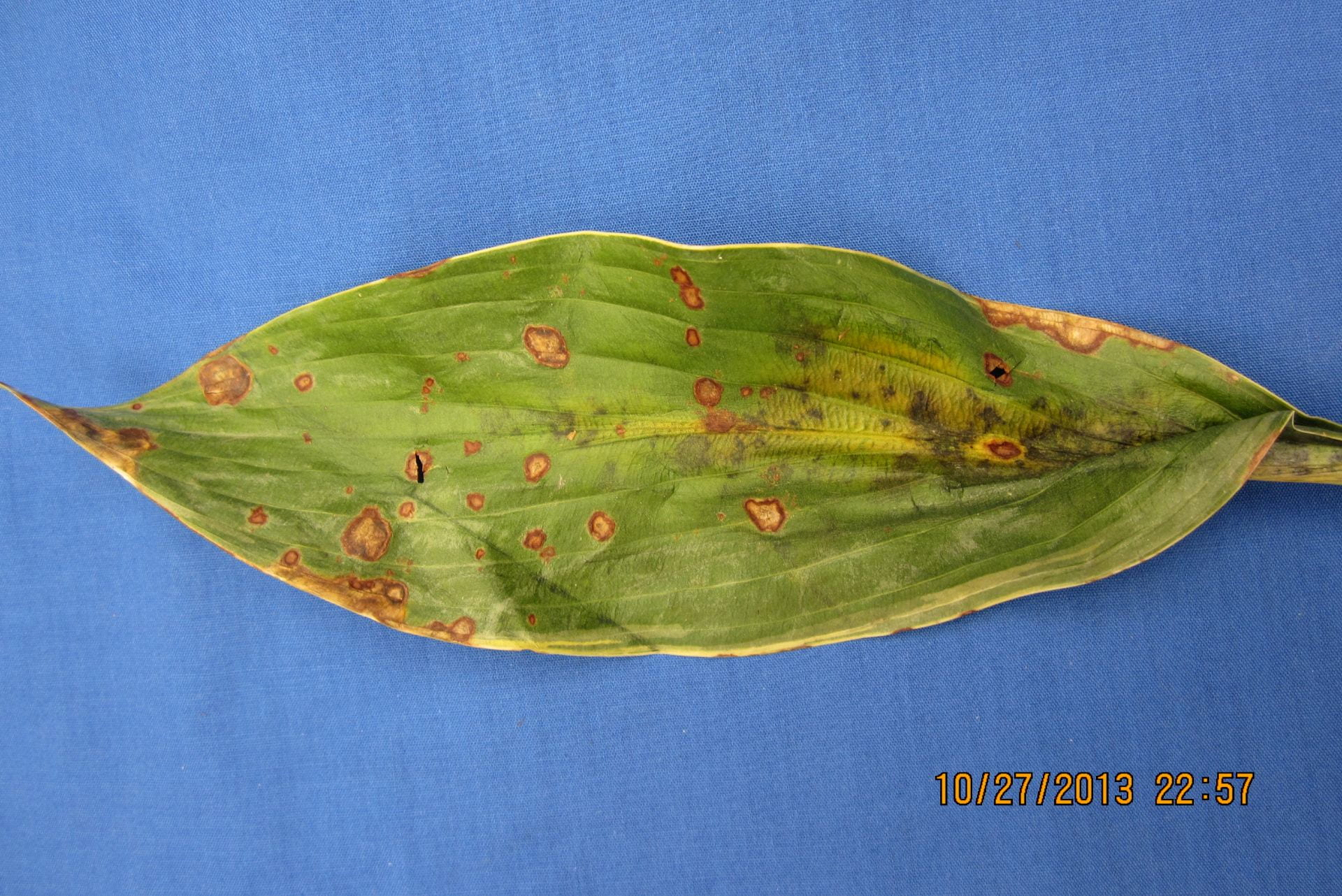
Dollar spot
Dollar spot in an untreated bentgrass area:

The photo above is a worse-case scenario, with a susceptible cultivar untreated. If you are having breakthroughs, review your fungicide regime and maybe your N regime, as low N can make this one worse (but too much is a problem, too!). Check your calibration and coverage. If you’ve never read it, read the dollar spot section starting on page 14 here:
http://www2.ca.uky.edu/agcomm/pubs/ppa/ppa1/ppa1.pdf
Unlike brown patch, dollar spot loves the more moderate temps of September, so this one does not fade away after Labor Day. So don’t let your guard down yet.
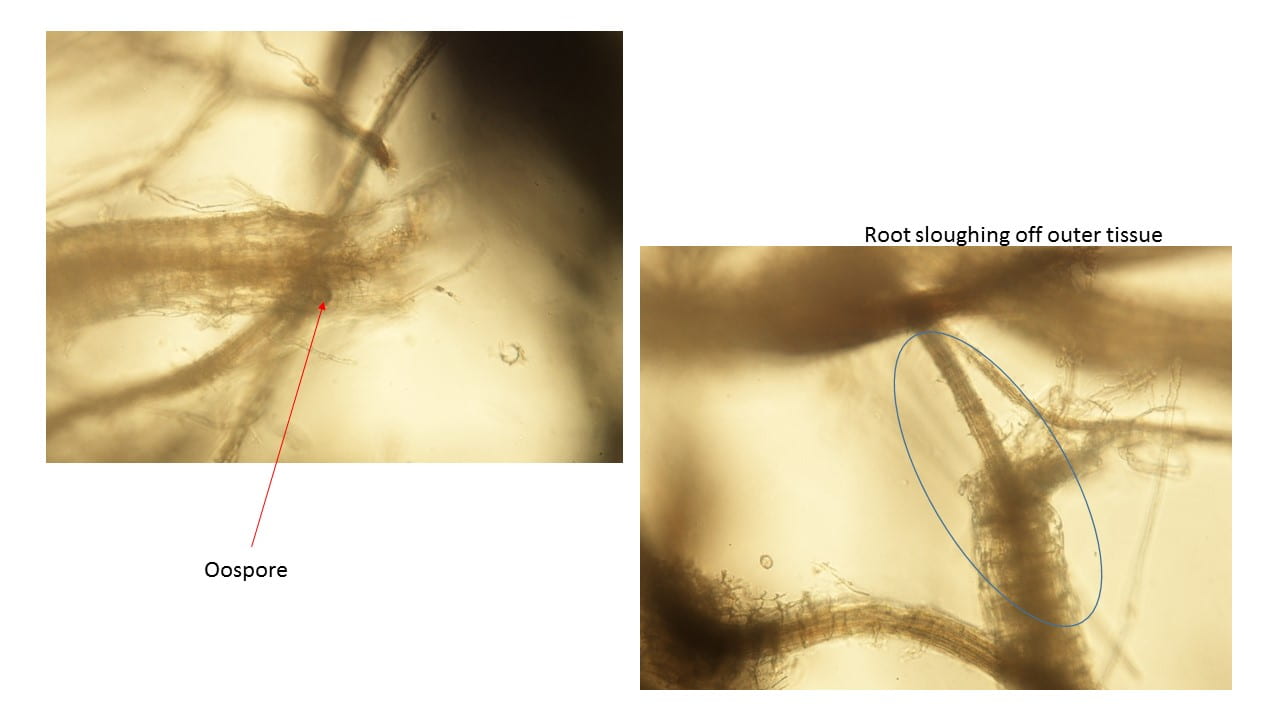
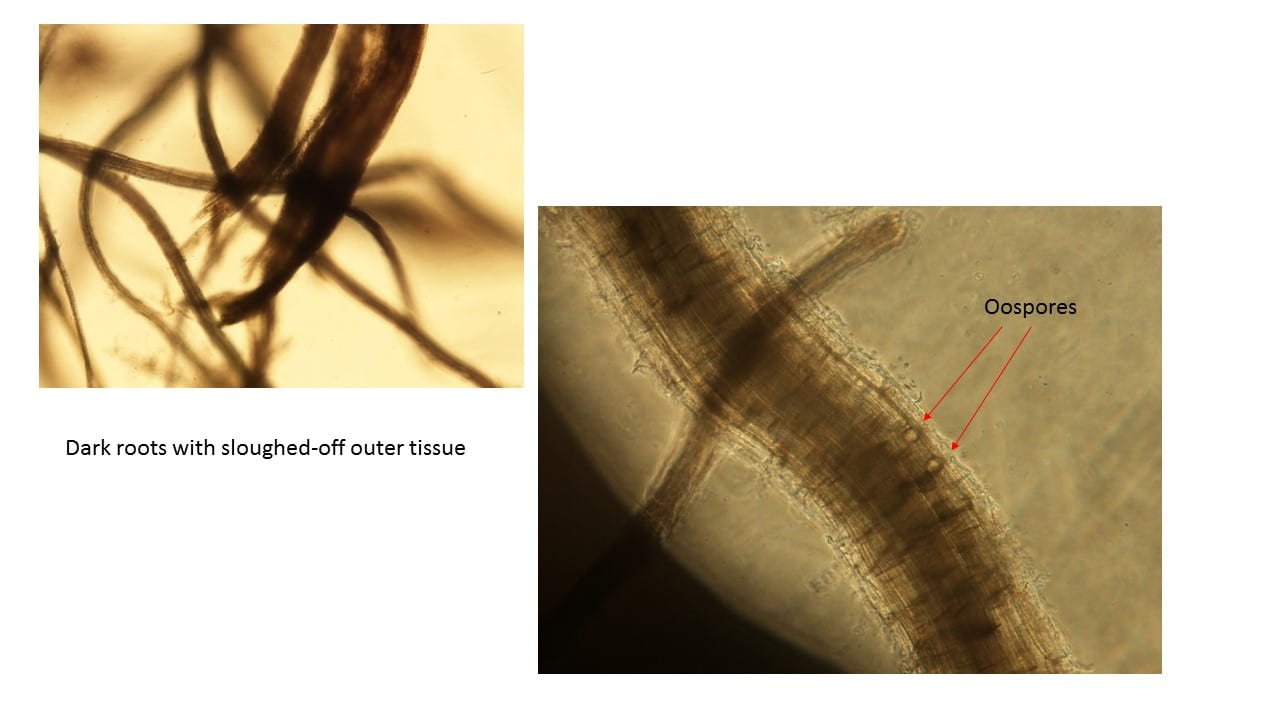
Continued root and crown woes
Another issue I’ve been seeing lately is continued problems with saturated roots, and there were more heavy rains this week. The wet soils deprive the roots of oxygen plus they hold heat, so turf does not have any “chill out time” overnight. Organic matter in the profile makes it worse. On top of the physiological issues, those wet soils can trigger Pythium root rot and sometimes anthracnose crown rot comes in on top of that.
For more details, you can check out the report this week from our great neighbor Dr. Miller here:
https://turfpath.missouri.edu/reports/2019/08_15_19/
Or here:
https://blogs.k-state.edu/turf/anthracnose-crown-rot-in-putting-greens-and-the-turf-stress-behind-it/
Localized dry spot
Finally, another issue I’d like to mention is hydrophobic soils. Despite all the rain, hydrophobic soils/localized dry spot can still occur.
Here is what localized dry spot can look like, and it can come on fast.


Keep an eye on your soil profile so you can stop this damage before it starts. When you get damage to the extent shown in those two photos above it is a long road to recovery.
To check for localized dry spot, pull up some cores and use the “droplet test” by putting drops of water on the plug. If the drops just sit there, not wicking in, you have a problem. The soil is water-repellent.


Sometimes there is a defined hydrophobic layer with normal soil above and/or below, so check at different depths, all the way down the rootzone:

You might also notice water beading up on the surface and not wicking in.
You don’t want this to sneak up on you. Keep an eye on your soil profile, especially locations with a history of problems. Aerification and use of wetting agents can help get moisture where it needs to go.